报告题目:Fluorophore Coupling Constrained by Multiple Cucurbit[8]uril Clampings:
from Discrete Dimers to Efficient Energy Transfer
报 告 人:Dr. Guanglu Wu
报告时间:2020年1月3日上午9:30 (星期五)
报告地点:无机超分子楼一楼圆形报告厅
Biography
Dr Guanglu Wu received his B.Sc. degree in Chemistry and completed his PhD degree under the supervision of Prof Xi Zhang at Tsinghua University, focusing on investigating the controlled self-assembly of bola-amphiphiles and its counterion effect. In December 2014, he joined Prof Oren Scherman’s group at the University of Cambridge as a postdoctoral research associate working on exploiting the dynamic aspect of supramolecular systems. His recent research revealed a neglected binding mode of CB[8]-mediated complexes, which offers a convenient way to fabricate discrete fluorophore dimers.
Abstract
Coupling two fluorophores within a sufficiently short distance for an extended period of time is crucial for both theoretical and experimental investigation of intermolecular processes. However, stacking together precisely two fluorophores in an aqueous solution remains a substantial challenge as most aromatic hydrocarbons show a tendency to aggregate unpredictably (forming clusters of arbitrary numbers of molecules).
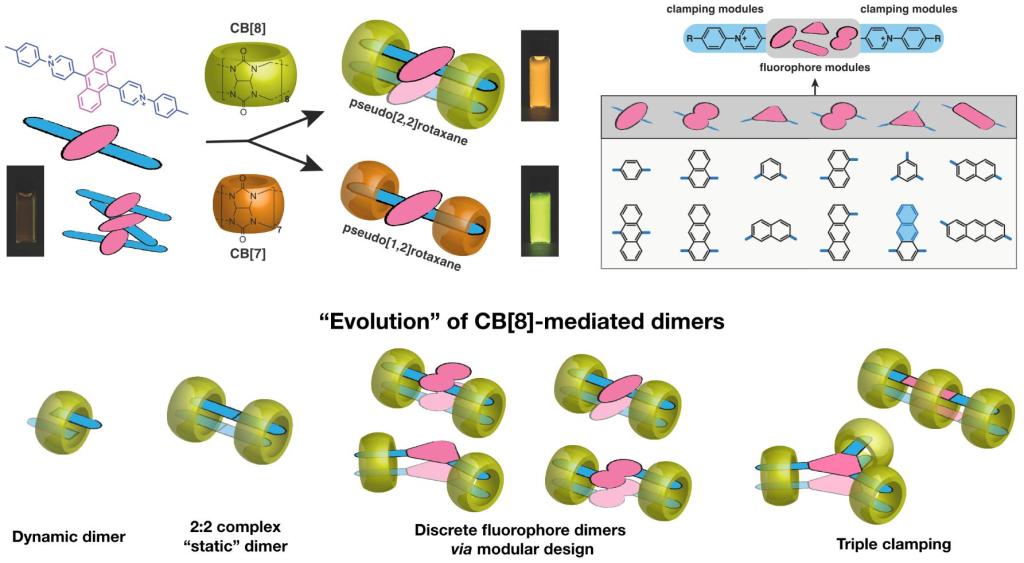
In 2017, diarylviologen derivatives with a direct connection of two arylpyridinium motifs were demonstrated to fabricate 2:2 complexes with cucurbit[8]uril (CB[8]) macrocycles, suggesting an effective way to confine two components to close proximity. Herein, by further using arylpyridinium as a clamping module, a new class of fluorescent molecules are developed, which generates discrete, dimeric stacked fluorophores upon complexation with multiple CB[8] macrocycles. The multiple constraints result in a "static" complex (remaining as a single entity for more than 30 ms) and facilitate fluorophore coupling in the ground state, showing a significant bathochromic shift in absorption and emission. This modular design is surprisingly applicable and flexible and has been validated through an investigation of nine different fluorophore cores ranging in size, shape, and geometric variation of their clamping modules. The resultant fluorescent dimers can be photo-excited to atypical excimer-like states with elongated excited lifetimes (up to 37 ns) and substantially high quantum yields (up to 1). This strategy offers a straightforward preparation of discrete fluorophore dimers, providing promising model systems with explicitly stable dimeric structures and tunable photophysical features, which can be utilized to facilitate various intermolecular processes.
References
1 Wu, G.; Olesińsk, M.; Scherman, O. A. et al. J. Am. Chem. Soc. 2017, 139, 3202.
2 Olesińsk, M.; Wu, G; Scherman, O. A. et al. Chem. Sci. 2019, 10, 8806.
3 Wu, G.; Scherman, O. A. et al. Chem. Comm. 2019, 55, 13227.
4 Wu, G; Scherman, O. A. et al. Chem. Sci. 2020, DOI: 10.1039/c9sc04587b.